Chapter
2
Understanding Ultraviolet Radiation
- Electromagnetic spectrum
- UVA, UVB and UVC
- Wave theory
- Quantum theory
In order to truly understand the tanning process of the skin you
need to have at least a basic understanding of the properties and
function of light. Although light has played a central role in the
histories of religion, art and science and is so common to our
everyday existance, it can actually be quite elusive.
Understanding Ultraviolet Radiation
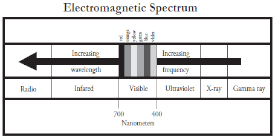
To understand ultraviolet radiation (UV) one needs to know UV�s placement in the
electromagnetic spectrum. Ultraviolet light is located between X-radiation and visible
light. UV has a higher frequency and shorter wavelength than visible light, and it
has a lower frequency and longer wavelength than X-radiation. UV with its longer
wavelength and less energy is less penetrating than X-ray and is sometimes absorbed
by matter. Photobiology studies the interaction of nonionizing radiation between the
electromagnetic spectrum and biologic systems. Nonionizing radiation represents the
ultraviolet, visible and near infrared regions of the spectrum. Tanning occurs as a
result of exposure to ultraviolet radiation. To fully understand this reaction, you must
familiarize yourself with the electromagnetic spectrum.
Electromagnetic Spectrum
The electromagnetic spectrum is a way of visualizing the frequency and wavelength
proportions of different forms of energy. Electromagnetic radiation has properties of
both waves and particles. We divide the electromagnetic spectrum in the UV range for
medical purposes.
UVA is found in the region between 320 and 400 nm (nm = Nanometer = 1 billionth
of a meter) and is the least powerful wavelength band of UV radiation. UVA acts
primarily to cause the melanin pigments in the skin to oxidize (darken) creating the
cosmetic tan and has limited power to cause erythema (sunburn).
UVB is found in the region between 280 and 320 nm. It comprises the wavelengths
primarily associated with erythema (sunburn), is also necessary for the production of
vitamin D in the skin and is primarily responsible for stimulating increased melanin
production. UVB wavelengths (at 305 nm) have 1,000 times more erythemal power
than UVA wavelengths.
UVC is found in the region between approximately 200-280 nm and is called
germicidal UV because of its proven effectiveness in killing single-cell organisms.
Solar radiation in the UVC range is absorbed almost entirely by the atmosphere and
that is fortunate considering that even a short overexposure to UVC is very harmful
to the eyes and causes severe erythema (sunburn). UVC is emitted by High Intensity
Discharge (HID) lamps; therefore these lamps require special filter glass, to contain
the output of the UVC spectrum (this will be discussed later). Another place where
radiation in the UVC range can be found is in the arc of a welding torch. For that
reason, optical damage referred to as �welders eye� is caused by UVC light.
Wave Theory
 Ultraviolet rays are similar to X-rays, white (visible) light, infrared and other similar
types of radiant energy. They are all electromagnetic waves, wavelike disturbances
associated with vibrating electric charges. Most waves are transmitted by some
medium; for example, you have all seen waves on the surface of the water, in which
case the water is the transmitting material. When a stringed instrument is plucked,
waves are set up in the string, so the string becomes the transmitting material.
Strangely enough, no one knows what transmits electromagnetic waves, however, we
have proof that they are in fact transmitted. Ultraviolet rays are similar to X-rays, white (visible) light, infrared and other similar
types of radiant energy. They are all electromagnetic waves, wavelike disturbances
associated with vibrating electric charges. Most waves are transmitted by some
medium; for example, you have all seen waves on the surface of the water, in which
case the water is the transmitting material. When a stringed instrument is plucked,
waves are set up in the string, so the string becomes the transmitting material.
Strangely enough, no one knows what transmits electromagnetic waves, however, we
have proof that they are in fact transmitted.
Electromagnetic waves all travel at the same constant speed as light, 186,000 miles
per second in a vacuum. All electromagnetic waves have the same form and travel
at the same speed, but differ in wavelength. Wavelength is the distance between
two successive crests in the wave. The number of crests or cycles per second is the
frequency of the wave. The unit of frequency is hertz or 1 cycle per second. Therefore,
if the wavelength is decreased, then the frequency is increased. Frequency and
wavelength have an inverse relationship which is calculated with one of two equations
where the velocity of radiation is 186,000 miles per second.
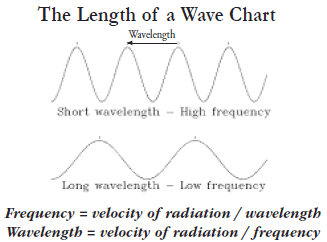
Where the velocity of radiation is 186,000 miles per second. Frequency is calculated using cycles per second and wavelength is calculated in meters. The wavelengths of electromagnetic radiation vary in size from a fraction of an angstrom unit (an angstrom is equal to ten billionths of a meter) to thousands of meters, commonly called the "electromagnetic spectrum." Some of the wavelengths of electromagnetic radiation from this spectrum are classified as follows:
| SPECTRUM |
APPROXIMATE WAVELENGTH
|
| X-Ray |
0.1 - 100 angstroms |
| Vacuum |
10 - 200 nanometers |
| Ultraviolet C (UVC) |
200 - 290 nanometers |
| Ultraviolet B (UVB) |
290 - 320 nanometers |
| Ultraviolet A (UVA) |
320 - 400 nanometers |
| Visible light |
400 - 700 nanometers |
| Near Infrared |
0.74 - 1.5 micrometers |
| Middle Infrared |
1.5 - 5.6 micrometers |
| Far Infrared |
5.6 - 1,000 micrometer |
| Microwave/Radiowaves |
greater than one millimeter |
Therefore, the useful unit of measure for our purposes is the nanometer. Radiations shorter than 10 nanometers (i.e. gamma rays or X-rays) generally ionize molecules (remove electrons) producing positively or negatively charged ions and are, therefore, known as ionizing radiation. Ultraviolet radiation is absorbed by molecules and is known as nonionizing radiation.
Quantum (Particle) Theory
Another theory used in reference to the electromagnetic spectrum is the quantum theory. In order to explain energy transfer, a bit of energy called a photon was theorized. Photons have no mass and when absorbed this energy is passed on to the absorbing molecule (such as skin cells) and the photon no longer exists in its same state. The amount of energy in a photon is directly proportional to the frequency of the radiation. The energy of a photon increases as the frequency increases. The more cycles per second (frequency) of any given photon, the more energy the photon has. The energy of any given photon decreases as the wavelength increases. The longer the wavelength, the less the frequency.
Light energy is expressed differently. We often express radiant energy in terms of watts per square meter or milliwatts per square centimeter. Skin exposure is usually expressed in joules per square centimeter. A Joule, is a unit of measurement and is equivalent to the electrical work done in one second by an electrical current of one ampere through the resistance of one ohm; named for its inventor, British Physicist, J.P. Joule (1818-1889).
|